
The inner orbitals surrounding the atom are spherical but the outer orbitals are much more complicated.Īn atom's electron configuration refers to the locations of the electrons in a typical atom. Today, this model is known as the quantum model or the electron cloud model. Electrons surround the atomic nucleus in pathways called orbitals, an idea that was put forth by Erwin Schrödinger, an Austrian physicist, in the 1920s. Originally known as "corpuscles," electrons have a negative charge and are electrically attracted to the positively charged protons. Joseph John (J.J.) Thomson, a British physicist, discovered the electron in 1897, according to the Science History Institute. Electrons are about 0.054% as massive as neutrons, according to Jefferson Lab. What is an electron?Įlectrons are tiny compared to protons and neutrons, over 1,800 times smaller than either a proton or a neutron. Three quarks make up each proton - two "up" quarks (each with a two-thirds positive charge) and one "down" quark (with a one-third negative charge) - and they are held together by other subatomic particles called gluons, which are massless. Elements are arranged in the Periodic Table of the Elements in order of increasing atomic number. The number of protons also determines the chemical behavior of the element. The number of protons in an atom is referred to as the atomic number of that element. For example, carbon atoms have six protons, hydrogen atoms have one and oxygen atoms have eight. The number of protons in an atom is unique to each element. Protons are about 99.86% as massive as neutrons according to the Jefferson Lab. Rutherford discovered them in experiments with cathode-ray tubes that were conducted between 19. Protons are positively charged particles found within atomic nuclei. These atoms will then decay into other elements, such as carbon-14 decaying into nitrogen-14. 
Some atomic nuclei are unstable because the binding force varies for different atoms based on the size of the nucleus. This force between the protons and neutrons overcomes the repulsive electrical force that would otherwise push the protons apart, according to the rules of electricity. The nucleus is held together by the strong force, one of the four basic forces in nature. The protons and neutrons that make up the nucleus are approximately the same mass (the proton is slightly less) and have the same angular momentum, or spin. Virtually all the mass of an atom resides in its nucleus, according to Chemistry LibreTexts. He also theorized that there was a neutral particle within the nucleus, which James Chadwick, a British physicist and student of Rutherford's, was able to confirm in 1932. In 1920, Rutherford proposed the name proton for the positively charged particles of the atom. The nucleus was discovered in 1911 by Ernest Rutherford, a physicist from New Zealand, according to the American Institute of Physics. Adding a proton to an atom makes a new element, while adding a neutron makes an isotope, or heavier version, of that atom. 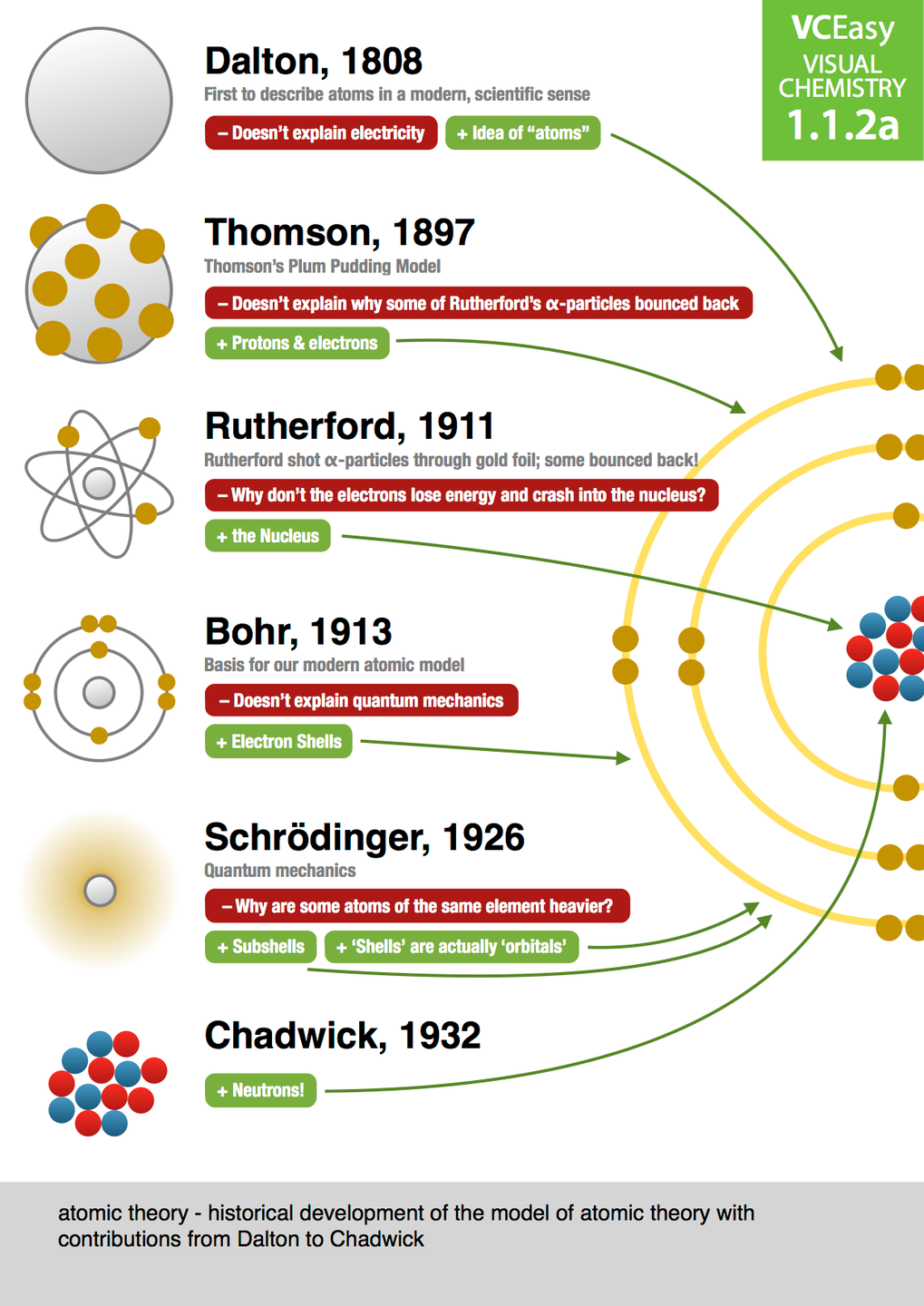
Atoms always have an equal number of protons and electrons, and the number of protons and neutrons is usually the same as well. However, one proton is about 1,835 times more massive than an electron. mass number (mass : charge ratio).Protons and neutrons have approximately the same mass. Isotopes are separated through mass spectrometry MS traces show the relative abundance of isotopes vs. There are naturally occurring isotopes and isotopes that are artificially produced. Isotopes Atoms that have the same atomic number (number of protons), but different mass numbers (number of protons and neutrons) are called isotopes.This is the energy required to remove an electron, in this case, from a molecule. Ionization Energies of Diatomic Molecule The energies of electrons in molecular orbitals can be observed directly by measuring the ionization energy.Lavoisier's Law of Conservation of Mass.His findings were based on experiments and the laws of chemical combination. Dalton's Atomic Theory John Dalton, a British school teacher, published his theory about atoms in 1808.Although the nucleus is heavy, it is quite small compared with the overall size of an atom. Most of an atom’s mass is in its nucleus the mass of an electron is only 1/1836 the mass of the lightest nucleus, that of hydrogen. The positive charges equal the negative charges, so the atom has no overall charge it is electrically neutral. Atomic Structure An atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.The atomic mass is used to find the average mass of elements and molecules and to solve stoichiometry problems. The mass of an atom or a molecule is referred to as the atomic mass. Atomic Mass Mass is a basic physical property of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
